- Blog
- About
- Contact
- Elysium Hindi Torrent
- Fem Simulation Companies In Germany
- Hemp Shipping Paper
- The Healer: Medieval Fantasy Romance By Dani Collins Epub
- Ok Google Vietnamese
- Alcatel One Touch Pop C5 5036d
- Iptv Shqip Pc
- Indian Chief 1932 Parts
- 3planetsoft Screensaver Serials
- Audio Story Of The Three Little Pigs
- Koyelaanchal Hindi Movie Download 720p
- Buku Makroekonomi Paul Samuelson Pdf
- Adobe Photoshop 7.0 Free Download Full Version With Key
- Nelly Discography Torrent
- Cuntwars Browser Hack
- Robotstudio 6 License Key
- Van Der Waals Forces In Graphite
- Pfense Wifi Internet Not Avilablae
- American Progress Painting
- Sell Any Watch Online
- Corel Paintshop Pro 2018 20.1.0.15 Portable
- Anytrans File Already Exists
- Ubuntu Route Add
- Hdd Mybook Essential 1tb Wd100001u
- Data Mining For Business Analytics
- Fl Studio 20.1.2.887 Patcher Full
- Fail2ban Proxmox Container
- Things That Add To Your Island Level In Skyblock
- Bangla Quran Tilawat Full 30 Para
- Savage Worlds Sci Fi Companion Pdf Download
- Warrior Cats Game On Scratch
- Mitsubishi Fx Simulator Use
- Wifislax 2017 Download
- Replay Video Capture
- Foto Di Windows 10 Tidak Bisa Dibuka
- May Allah Bless You In This World And Hereafter
- Taylormade M4 Hybrid
- Xerox Cp6334 Driver
- Hack Whatsapp Number
- Hard Drive Defragger
- Ciclo For En Php Formulario
- Gtk 2.24 Libraries
Archived
Are graphene layers in graphite bonded through Van Der Waals or weak 'metallic' bonds?
May 9, 2017 - Graphite is composed of stacked layers of graphene sheets, which are held together by the weak Van der Waals forces, including attraction. Van der Waals bonding is common in gases and organic liquids and solids, but it is rare in minerals. Its presence in a mineral defines a weak area with good cleavage and low hardness. In graphite, carbon atoms lie in covalently bonded sheets with van der Waals forces acting between the layers. Hydrogen bonds.
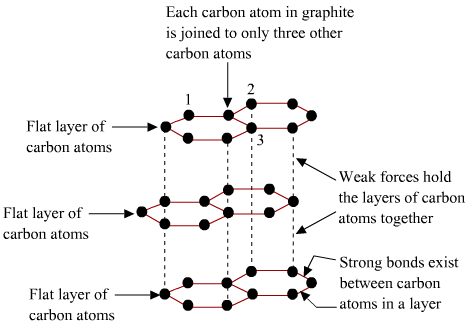
I was always taught, and numerous sources stipulate, that the graphene layers are held together in graphite through long range Van Der Waals attraction force between the graphene layers. However, I just read on a paper that this view is erroneous due to the fact that graphite exhibits interlayer current; indeed, this would cause graphite to have a more 'metallic' bond that allows electron flow throughout the entire molecular structure. The paper was dated 2007; is this the current view of the bonding in graphite?
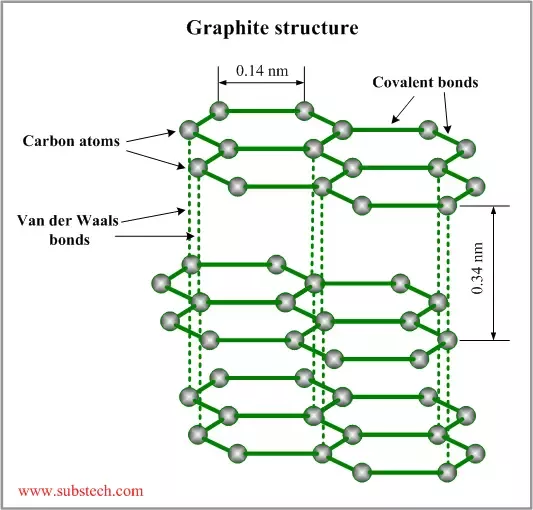
Link to the paper: http://przyrbwn.icm.edu.pl/APP/PDF/112/a112z308.pdf
78% Upvoted
